
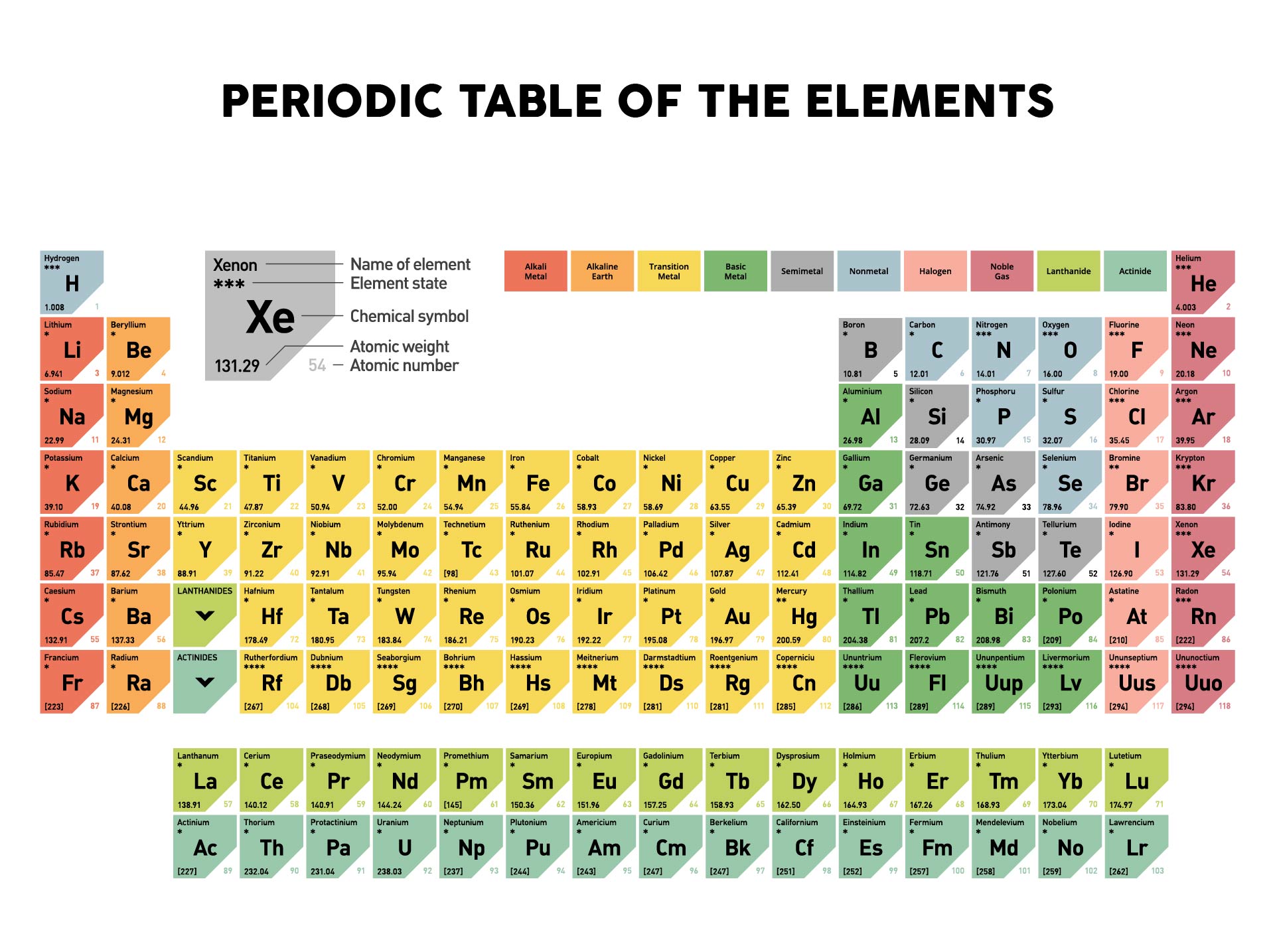
You will need to refer to a periodic table for proton values. This periodic table is suitable for a computer and mobile device wallpaper. The IUPAC doesnt update these values annually, so these are the most recent values for 2019. The table also includes electronegativity values. Average molar masses are rounded to two decimal places.

To view and print these files, you will need to install the free Adobe Acrobat Reader program on your computer. In this notation, the atomic number is not included. This color periodic table contains the accepted standard atomic weights (atomic masses) of each element as accepted by the IUPAC. Unless otherwise indicated, the following tables are in Acrobat PDF format. The atomic mass is carried by the atomic nucleus, which occupies only about 10 -12 of the total volume of the atom or. Symbol-mass format for the above atom would be written as Cr-52. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. The "A" value is written as a superscript while the "Z" value is written as a subscript. Both the atomic number and mass are written to the left of the chemical symbol. The molar mass of a substance ( M) is the weight of one mole of the substance. Fe4 Fe (CN)63, NaHCO3, ch3coonh4, h2so4, pb (c2h3o2)23h2o, caso41/2h2o) and press Enter or click Calculate button. The composition of any atom can be illustrated with a shorthand notation called A/Z format. Enter a chemical formula to calculate its molar mass (e.g. Thus, the molar mass of bilirubin can be expressed as 584.05 g/mol, which is read as “five hundred eighty four point zero five grams per mole.\nonumber \] The division sign (/) implies “per,” and “1” is implied in the denominator. So, in our example, carbon has a molar mass of 12.01 grams per mole. For example, if you want to find the molar mass of carbon, you would find the atomic mass of carbon on the periodic table, and this is equal to the molar mass in grams per mole. For example, the molar mass of Ba(OH) 2 requires the sum of 1 mass of Ba, 2 masses of O, and 2 masses of H: 1 Ba molar mass:īecause molar mass is defined as the mass for 1 mol of a substance, we can refer to molar mass as grams per mole (g/mol). The molar mass of elements is found by looking at the atomic mass of the element on the periodic table. In formulas with polyatomic ions in parentheses, the subscript outside the parentheses is applied to every atom inside the parentheses. The mass of 1 mol of bilirubin is 584.69 g.īe careful when counting atoms. Using the periodic table to find the mass for each mole of our elements we have: 1moleC 1 moleC ×(12.011gC 1 moleC) 12.011gC (8.3.8) (8.3.8) 1 m o l e C 1 m o l e C × ( 12.011 g C 1 m o l e C) 12.011 g C. Multiplying the molar mass of each atom by the number of atoms of that type in bilirubin’s formula and adding the results, we get First we need to determine the mass of one mole of methane (CH 3 OH).Summing the molar masses of the atoms in the NaCl formula unit gives The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. bilirubin (C 33H 36N 4O 6), the principal pigment present in bile (a liver secretion).
What is the mass of 1 mol of each substance? \): Moles to Mass Conversion with Compounds The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element.
